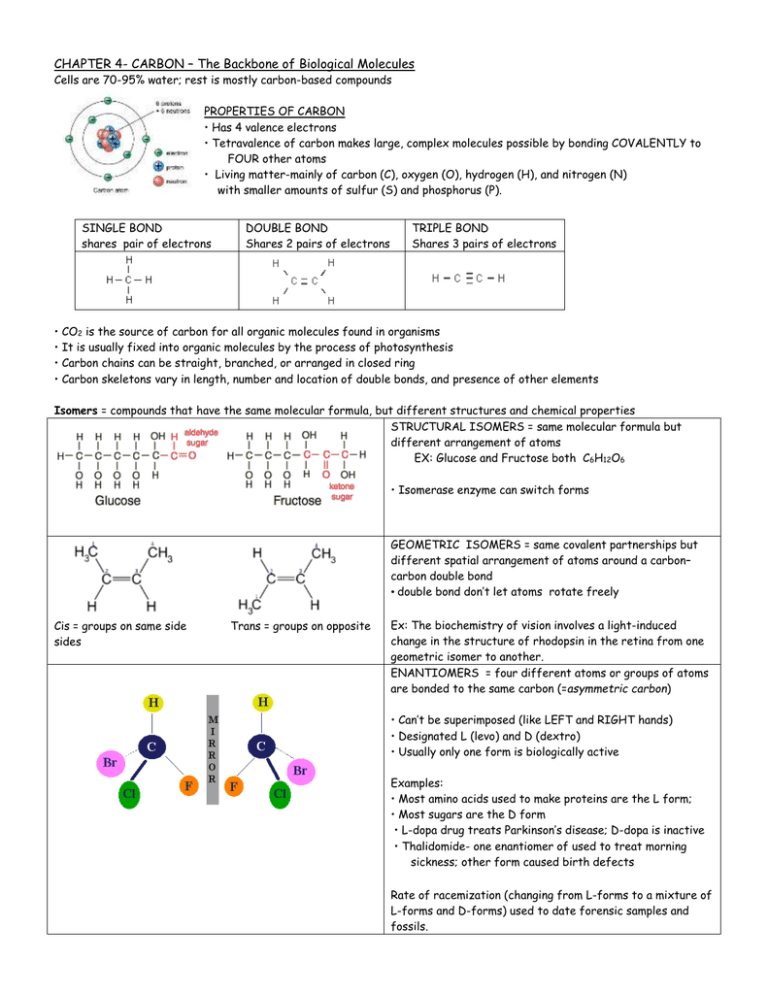
Can you identify any of the common functional groups on the following biological molecules?ģ9. Can you identify any of the common functional groups on the following biological molecules? Starch and celluloseStarch and celluloseģ8. A small number of chemical functional groups are key to the function of biological moleculesģ7. A small number of chemical functional groups are key to the function of biological molecules Properties of the molecule will also depend on the molecular components attached to the carbon skeletonFunctional groups can replace H These groups participate in chemical reactions and change the molecular shapeģ6. A small number of chemical functional groups are key to the function of biological molecules We’ve been focusing on the carbon skeleton….what’s attached impacts form and functionWe’ve been focusing on the carbon skeleton….what’s attached impacts form and functionģ5. Other forms of diversity Three types of isomersStructuralGeometricEnantiomers are isomers that are mirror images of each otherģ4. Other forms of diversity Three types of isomersStructuralGeometric isomers have the same covalent partnerships, but differ in their spatial arrangement (related to inflexibility of double bonds)ģ3. Other forms of diversity Three types of isomersStructuralGeometric isomers have the same covalent partnerships, but differ in their spatial arrangement (related to inflexibility of double bonds)ģ2. Other forms of diversity Three types of isomersStructural isomers differ in the arrangement of their atoms or in the presence or absence of double bondsģ1. Other forms of diversity Three types of isomersStructural isomers differ in the arrangement of their atoms or in the arrangement of double bondsģ0. Other forms of diversity Three types of isomersStructural isomers differ in the arrangement of their atoms or in the arrangement of double bondsĢ9. Other forms of diversity Three types of isomersStructural isomers differ in the arrangement of their atoms or in the arrangement of double bondsĢ8. 
Other forms of diversity Three types of isomersStructuralGeometricEnantiomersĢ7. Other forms of diversity Isomers-compounds that have the same numbers of atoms of the same elements, but different structures (hence different properties)Ģ6. Molecular diversity arising from carbon skeleton variationĢ5. Molecular diversity arising from carbon skeleton variation Sources of diversity:Chain lengthBranchingDouble bondsRing formationĢ3. Molecular diversity arising from carbon skeleton variation Sources of diversity:Chain lengthBranchingDouble bondsĢ2. Molecular diversity arising from carbon skeleton variation Sources of diversity:Chain lengthBranchingDouble bondsĢ1. Molecular diversity arising from carbon skeleton variation Sources of diversity:Chain lengthBranching, ,Ģ0. Molecular diversity arising from carbon skeleton variation Sources of diversity:Chain lengthġ9. Molecular diversity arising from carbon skeleton variation Sources of diversity:Chain lengthġ8. Molecular diversity arising from carbon skeleton variation Sources of diversity:Chain lengthġ7. Molecular diversity arising from carbon skeleton variationġ6. Carbon atoms can form diverse molecules by bonding to four other atomsġ5. Carbon atoms can form diverse molecules by bonding to four other atomsġ4. Carbon atoms can form diverse molecules by bonding to four other atoms Carbon usually completes its valence shell by sharing electrons with other atoms in covalent bondsġ3. Carbon atoms can form diverse molecules by bonding to four other atoms Carbon usually completes its valence shell by sharing electrons with other atoms in covalent bondsġ2. Carbon atoms can form diverse molecules by bonding to four other atoms Carbon usually completes its valence shell by sharing electrons with other atoms in covalent bonds. Carbon atoms can form diverse molecules by bonding to four other atoms Carbon usually completes its valence shell by sharing electrons with other atoms in covalent bondsġ0. Carbon atoms can form diverse molecules by bonding to four other atoms The key to an atom’s chemical characteristics is its electron configuration Carbon’s valence shell is half-full…or is it half-empty?ĩ. Carbon atoms can form diverse molecules by bonding to four other atomsĨ. Organic chemistry is the study of carbon compounds It mostly involves CHOPNSWith few elements it is possible to create a diversity of molecules….because of the special properties of carbonħ. Organic chemistry is the study of carbon compounds Exception:Ħ. Organic chemistry is the study of carbon compounds The term “organic” chemistry came from the misconception that carbon-based compounds were always connected to life Exception:ĥ. Organic chemistry is the study of carbon compounds The term “organic” chemistry came from the misconception that carbon-based compounds were always connected to lifeĤ.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
